New federal hemp definitions
The 2026 federal definition of hemp is moving away from the old 0.3% delta-9 THC dry weight limit. Instead, the government is switching to a 'total THC' calculation. This includes delta-8 THC in the final percentage, which effectively lowers the legal threshold for what qualifies as hemp.
Since delta-8 is usually synthesized from CBD, this change hits the market hard. Products that were legal yesterday might be over the limit tomorrow. I've seen growers scrambling to change their testing protocols, but many are still struggling to figure out if their current crops are even legal under the new math.
The shift to a total THC calculation is intended to close a loophole that allowed for the proliferation of intoxicating hemp-derived products. While proponents argue this protects consumers, it's creating significant disruption in the industry. It presents challenges for accurately testing products and ensuring consistent compliance across the supply chain. The change also complicates interstate commerce, as products legal in one state might be illegal in another due to varying interpretations of the new federal rule.
I've heard from several smaller CBD producers who are deeply concerned about the costs associated with re-testing their existing inventory and modifying their manufacturing processes. Larger companies with more resources will likely adapt more easily, potentially creating an uneven playing field. It’s a situation that requires careful monitoring and advocacy to ensure a fair and sustainable CBD market.

The FDA's 2026 outlook
The Food and Drug Administration (FDA) continues to hold a cautious position on CBD, as outlined in their official statements on fda.gov. Their primary concerns center around safety – a lack of sufficient data on long-term effects – and inconsistent labeling practices. The FDA has issued warning letters to companies making unsubstantiated health claims about CBD and has taken enforcement actions against those selling adulterated or misbranded products.
Currently, the FDA maintains that CBD cannot be legally marketed as a dietary supplement or food additive without further research and approval. They argue that adding CBD to food introduces a drug into the food supply, which requires a different regulatory pathway. This stance has created a gray area for many CBD companies, forcing them to navigate a complex web of regulations and potential legal risks.
Looking ahead to 2026, predicting the FDA’s actions is difficult, but the congressional updates regarding the hemp definition might prompt some movement. I suspect we’ll see increasing pressure on the FDA to establish a clear regulatory framework for CBD, especially if the agency continues to receive data demonstrating the safety of certain CBD products. A potential pathway for CBD as a food additive or dietary supplement could emerge, but it will likely be accompanied by strict regulations regarding dosage, labeling, and quality control.
However, substantial hurdles remain. The FDA requires extensive data to demonstrate the safety of CBD, and conducting these studies is expensive and time-consuming. There’s also the question of whether the FDA will differentiate between various CBD product categories – for example, applying stricter rules to ingestible products than to topicals. It's not a simple process, and I think it's realistic to expect continued uncertainty for the foreseeable future. The agency needs more data, and the industry needs clarity.
The FDA has shown a willingness to work with Congress on potential solutions, but their commitment to consumer safety remains paramount. Enforcement actions will likely continue, particularly against companies that disregard existing regulations or make misleading claims. Businesses should prioritize compliance and transparency to avoid potential legal issues.
- The FDA is sending warning letters to brands making health claims.
- FDA takes enforcement actions against companies selling adulterated or misbranded products.
- FDA requires extensive data to demonstrate the safety of CBD for various applications.
The state law mess
Even with the updated federal definition of hemp, state laws remain a significant factor in the CBD landscape. The new federal guidelines don’t automatically override existing state regulations, leading to a continued patchwork of laws across the country. This creates a complex challenge for businesses, especially those operating in multiple states.
States like New York and California have historically adopted stricter regulations on CBD, often requiring extensive testing, licensing, and labeling requirements. New York, for example, has implemented detailed rules regarding the sale of hemp-derived cannabinoid products, including restrictions on certain ingredients and marketing practices. California’s regulations are similarly stringent, with a focus on protecting consumers from potentially harmful products.
Conversely, states like Colorado and Oregon have generally taken a more permissive approach to CBD regulation, recognizing the economic benefits of the hemp industry. These states often have less restrictive licensing requirements and allow for a wider range of CBD products to be sold. This difference in approach creates opportunities for businesses to thrive in certain states while facing significant hurdles in others.
For CBD stores with multiple locations, navigating this patchwork of regulations is a logistical nightmare. They must ensure compliance with the laws of each state in which they operate, which can require significant investment in legal counsel and compliance personnel. The lack of uniformity also creates challenges for interstate commerce, making it difficult to efficiently distribute products across state lines.
State CBD Regulations - A Comparative Overview (as of late 2023/early 2024)
| State | Hemp-Derived CBD Legality | THC Limit | CBD in Food/Beverages | Medical CBD Program |
|---|---|---|---|---|
| Colorado | Fully Legal | ≤ 0.3% THC | Legal with restrictions (clear labeling required) | Established, comprehensive |
| California | Legal with restrictions | ≤ 0.3% THC | Legal with restrictions (complex regulations, requires FDA compliance) | Established, comprehensive |
| Texas | Legal with restrictions | ≤ 0.3% THC | Illegal (limited exceptions for topical applications) | Limited; Low-THC cannabis program for specific medical conditions |
| Florida | Legal with restrictions | ≤ 0.3% THC | Legal with restrictions (subject to Department of Agriculture and Consumer Services rules) | Established, comprehensive |
| New York | Legal with restrictions | ≤ 0.3% THC | Legal with restrictions (strict labeling and testing requirements) | Established, comprehensive |
| Idaho | Restricted | ≤ 0.3% THC (but with legal ambiguity) | Generally Illegal | No comprehensive medical CBD program |
| Illinois | Legal with restrictions | ≤ 0.3% THC | Legal with restrictions (regulated by the Department of Agriculture) | Established, comprehensive |
Illustrative comparison based on the article research brief. Verify current pricing, limits, and product details in the official docs before relying on it.
Impact on Global CBD Stores
The evolving CBD regulations have a direct impact on global CBD stores, particularly those sourcing products from different countries. The updated federal definition of hemp and the FDA’s stance on CBD create new compliance challenges for businesses importing or exporting CBD products. Ensuring that products meet both US federal regulations and the regulations of the exporting country is crucial.
Certificates of Analysis (COAs) from independent, third-party laboratories are now more important than ever. These COAs verify the THC content and purity of CBD products, providing evidence of compliance with regulatory requirements. Stores should prioritize sourcing products from reputable suppliers who provide comprehensive COAs and demonstrate a commitment to quality control.
Online CBD stores face additional challenges related to shipping restrictions. Major shipping carriers have varying policies regarding the shipment of CBD products, and some states prohibit the shipment of CBD altogether. Stores must carefully research and comply with these shipping regulations to avoid potential legal issues. It's a constant moving target, and staying informed is key.
We're updating our directory to reflect these 2026 changes. My team is manually verifying COAs for every listed store to make sure they aren't selling products that fall into this new 'total THC' trap. It's a lot of work, but it's the only way to keep the directory useful.
Stores need to be diligent in staying informed about changes in both federal and state regulations. Subscribing to industry newsletters, attending conferences, and consulting with legal counsel are all valuable strategies for maintaining compliance. Ignoring these changes could result in significant penalties, including fines, product seizures, and even criminal charges.
- Only buy from suppliers who provide full lab results (COAs).
- Stay informed about changing federal and state regulations.
- Comply with shipping restrictions for online CBD sales.
Banking and Financial Services
The banking industry has historically been hesitant to work with CBD businesses due to the federal illegality of cannabis. This reluctance has created significant challenges for CBD companies, limiting their access to essential financial services like checking accounts, loans, and credit card processing. Many CBD businesses have been forced to operate on a cash-only basis, which is risky and inefficient.
The new regulations, while not fully legalizing CBD at the federal level, could lead to some easing of banking restrictions. If banks perceive a reduced risk due to the updated hemp definition, they may be more willing to offer accounts to CBD companies. However, many banks remain cautious, awaiting further clarification from regulators.
There’s ongoing discussion about federal legislation to address banking issues specifically. The SAFE Banking Act, which would protect financial institutions that provide services to legitimate cannabis businesses, has gained traction in Congress but has yet to be passed. Its passage would likely have a significant positive impact on the CBD industry, providing greater access to banking services.
Currently, CBD businesses are using various workarounds to access financial services, such as partnering with specialized payment processors or utilizing offshore banking solutions. However, these options often come with higher fees and increased complexity. A more stable and accessible banking system is critical for the long-term growth of the CBD industry.
Rules for specific products
The regulatory landscape for CBD products isn’t uniform. Different product categories may be subject to varying levels of scrutiny. Ingestible products – oils, tinctures, edibles – generally face stricter regulations than topicals, due to the potential for systemic effects. The FDA has been particularly focused on ensuring the safety and accurate labeling of ingestible CBD products.
CBD cosmetics also present unique regulatory challenges. The FDA regulates cosmetics under the Federal Food, Drug, and Cosmetic Act, and requires that cosmetic products be safe and properly labeled. However, the agency’s stance on CBD in cosmetics remains unclear, and companies must navigate a complex web of regulations to ensure compliance. Labeling requirements for CBD cosmetics are particularly stringent, requiring accurate disclosure of ingredients and potential allergens.
A key debate centers around the classification of CBD as a dietary supplement versus a drug. If the FDA were to allow CBD to be marketed as a dietary supplement, it would open up new opportunities for the industry. However, this would require demonstrating the safety of CBD for long-term consumption and establishing clear quality control standards. The agency has previously indicated that CBD cannot be considered a dietary supplement without further research, and that position hasn’t changed significantly.
CBD Product Regulation Challenges – 2026
- CBD Oils & Tinctures - Continued scrutiny surrounds potency claims and the accurate labeling of total THC content, even with the 2018 Farm Bill’s stipulations. The FDA is expected to release further guidance on manufacturing practices, particularly regarding carrier oil quality and consistency, impacting sourcing for global stores.
- CBD Edibles (Gummies, Chocolates) - A major challenge remains the enforcement of rules against marketing edibles to children. New regulations are anticipated to clarify permissible ingredients and shapes, aiming to differentiate CBD edibles from conventional confectionery products. Global stores will need to verify supplier compliance with these evolving standards.
- CBD Topicals (Creams, Balms) - The FDA is focusing on the categorization of CBD topicals – are they cosmetics, drugs, or something else? This impacts labeling requirements and permissible health claims. Global stores should anticipate stricter guidelines on advertising benefits like pain relief or inflammation reduction.
- CBD Vape Products - Following concerns about the 2019 EVALI outbreak, vape products face the most stringent regulations. Expect increased requirements for testing for heavy metals, Vitamin E acetate, and other contaminants. Global stores sourcing vape products will need to demonstrate robust quality control measures.
- CBD Pet Products - The FDA is actively investigating the safety and efficacy of CBD in animal products. Regulations are expected to address appropriate dosages for different animal species and labeling requirements to prevent misleading claims. Global stores selling pet CBD will need to ensure products have undergone veterinary review or adhere to emerging standards.
- CBD Isolates & Distillates - While generally considered less regulated than full-spectrum products, the purity and source of isolates and distillates are coming under increased scrutiny. Expect more detailed requirements for Certificate of Analysis (COA) reporting and testing methodologies to verify cannabinoid content and the absence of harmful substances.
- Full & Broad Spectrum CBD Products - Maintaining legally compliant THC levels (below 0.3% Delta-9 THC) remains a key challenge. New testing protocols may be implemented to ensure accurate measurement of total THC, including Delta-8 THC and other cannabinoids. Global stores will need to rely on reliable testing labs and supplier documentation.




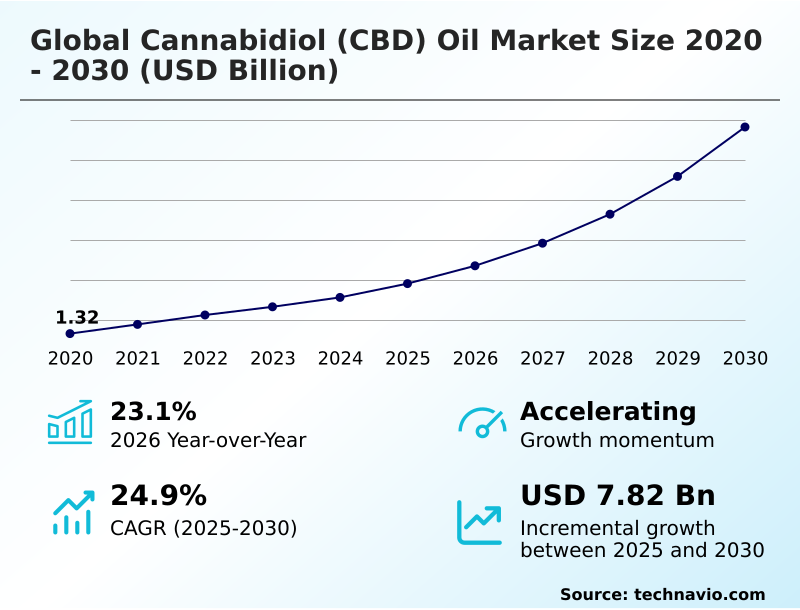
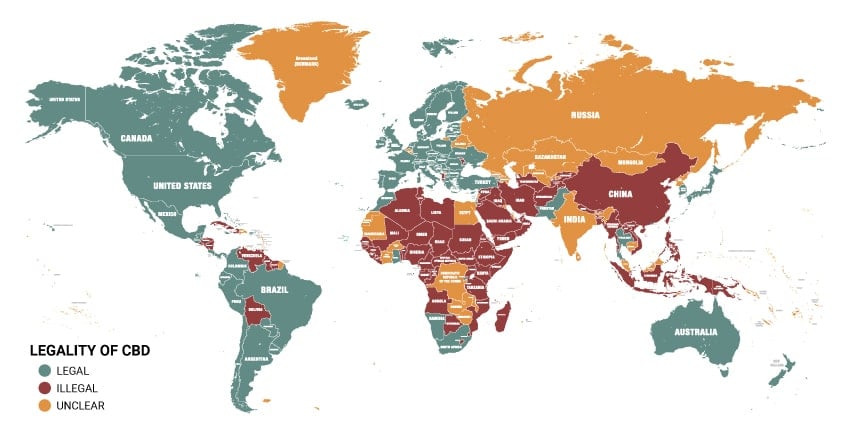
No comments yet. Be the first to share your thoughts!