Global CBD Store Growth: A Visual Overview
The global CBD market is experiencing a period of rapid expansion, and that’s visibly playing out in the number of places you can actually buy CBD. We’re seeing a surge in both brick-and-mortar stores and online retailers dedicated to, or including, CBD products. It’s not just in places you’d expect either – the growth is surprisingly widespread.
This isn’t just about a few isolated successes. The increase in global CBD stores reflects a broader shift in public perception. Acceptance is growing, and as it does, so does the demand. Changing regulations, albeit slowly and unevenly, are also a major factor, opening up new opportunities for businesses. It's a dynamic time, with new stores appearing frequently.
It's difficult to pinpoint an exact number for global CBD stores, as tracking is fragmented. However, anecdotal evidence and early market reports suggest a substantial increase over the past five years, and that trend is expected to continue. The recent surge in listings on platforms like MindCBD, our global CBD store directory, clearly illustrates this expansion. We're constantly adding new locations as they come online.
The growth isn't uniform. North America and Europe currently lead the way in terms of store density, but we’re seeing increased activity in Asia-Pacific and South America. This early expansion is setting the stage for what we expect to be even more significant growth in the coming years.
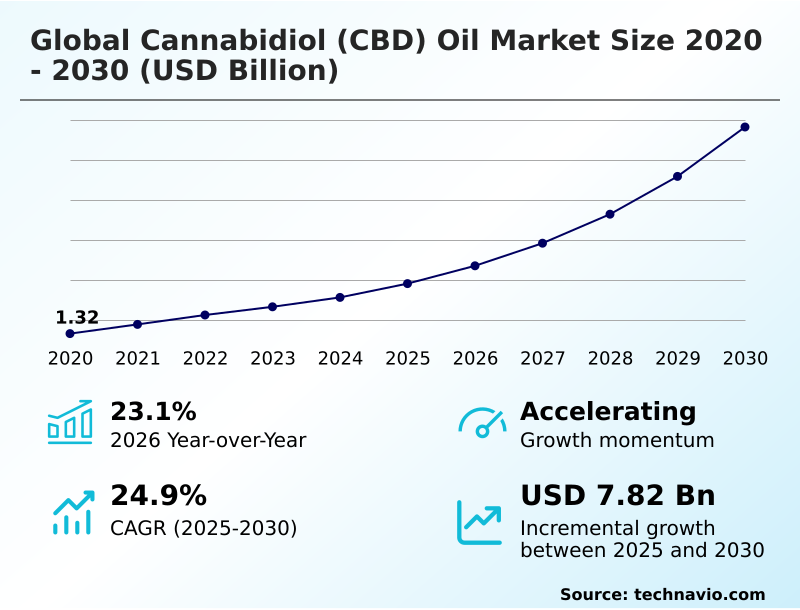
2026 Market Projections: Key Growth Regions
Market analysts are projecting continued robust growth for the global CBD market through 2026. Estimates vary, but most forecasts place the global CBD market size between $40 billion and $55 billion by 2026, up from around $27.5 billion in 2023. This growth won’t be evenly distributed, though.
Europe is anticipated to be a major driver of growth, with a projected market value of approximately $18 billion by 2026. This is largely fueled by the ongoing, albeit slow, harmonization of regulations within the European Union. Countries like the UK, Germany, and Italy are leading the charge, but even smaller markets are showing significant potential.
North America – specifically the United States and Canada – will remain a substantial market, estimated at around $15 billion by 2026. Growth here is hampered by federal uncertainty in the US, but state-level innovation continues to push the market forward. Canada, with its more established regulatory framework, is expected to see consistent growth.
The Asia-Pacific region is poised for explosive growth, with projections reaching $8 billion by 2026. This is driven by a combination of factors, including increasing acceptance of CBD for wellness purposes and the integration of CBD into traditional medicine practices. Key markets include Japan, South Korea, Australia, and Thailand.
South America is a bit of a wildcard. Regulatory frameworks are still developing, but the region has a large population and a growing interest in natural health products. Market projections for South America are more conservative, around $3 billion by 2026, but the potential for rapid growth is significant.
- Europe: Projected $18 billion by 2026 – driven by EU harmonization.
- North America: Projected $15 billion by 2026 – US federal uncertainty a limiting factor.
- Asia-Pacific: Projected $8 billion by 2026 – fueled by traditional medicine acceptance.
- South America: Projected $3 billion by 2026 – regulatory development underway.
Projected Global CBD Market Size & Growth (2023-2026)
| Region | Projected Market Size (2026 - USD Billions) | Projected CAGR (2023-2026) | Key Market Drivers |
|---|---|---|---|
| North America | Approximately $23.9 Billion | 21.4% | Established regulatory frameworks in some states/provinces, high consumer awareness, and product innovation. |
| Europe | Approximately $18.7 Billion | 19.8% | Increasing acceptance of CBD for wellness, growing retail presence, and evolving regulatory landscapes. |
| Asia-Pacific | Approximately $8.2 Billion | 26.7% | Rising disposable incomes, increasing health consciousness, and potential for significant growth in countries like Japan and South Korea. |
| Latin America | Approximately $3.1 Billion | 24.2% | Expanding access to CBD products, particularly in Mexico and Brazil, and increasing interest in alternative therapies. |
| Rest of World | Approximately $4.5 Billion | 22.1% | Growth driven by emerging markets in Africa, the Middle East, and other regions with developing CBD regulations. |
Illustrative comparison based on the article research brief. Verify current pricing, limits, and product details in the official docs before relying on it.
European Union: Harmonization and Hurdles
The European Union presents a unique challenge for CBD businesses. While there’s a clear appetite for CBD products across member states, the regulatory landscape is incredibly fragmented. Each country has its own rules regarding CBD production, sale, and THC content limits, creating a complex web for businesses to navigate.
The EU is attempting to harmonize these regulations through the Novel Food regulations. This requires CBD products to be authorized as novel foods before they can be legally sold. The process is lengthy and expensive, and has created significant hurdles for many businesses. It's also led to some uncertainty about the legal status of CBD products that were already on the market before the regulations came into effect.
One major obstacle to harmonization is the differing interpretations of EU law by individual member states. Some countries take a more lenient approach, allowing for the sale of broad-spectrum and full-spectrum CBD products, while others are much more restrictive, only permitting the sale of CBD isolates. This inconsistency creates confusion for both consumers and businesses.
A harmonized regulatory framework would undoubtedly accelerate market expansion in the EU. It would reduce compliance costs for businesses, create a level playing field, and provide greater clarity for consumers. It would also likely attract more investment into the European CBD market. However, achieving this harmonization is proving to be a slow and politically sensitive process.
The European Commission is currently reviewing the Novel Food regulations, and there's hope that changes will be made to streamline the authorization process. Some stakeholders are advocating for a more risk-based approach, focusing on product safety rather than simply requiring pre-market authorization. The outcome of this review will be crucial for the future of the CBD market in Europe.
- Novel Food Regulations: Require pre-market authorization, creating hurdles for businesses.
- Inconsistent THC Limits: Varying rules across member states create confusion.
- Differing Interpretations of EU Law: Lead to fragmented regulatory approaches.
- Ongoing Review of Novel Food Regulations: Potential for streamlining the authorization process.
United States: Federal Uncertainty and State-Level Innovation
The US CBD market is a behemoth, but its growth is significantly constrained by federal prohibition. While the 2018 Farm Bill legalized hemp-derived CBD containing less than 0.3% THC, the Drug Enforcement Administration (DEA) still maintains that CBD is a Schedule I controlled substance. This creates a legal gray area and complicates interstate commerce.
According to the U.S. Customs and Border Protection (cbp.gov), the import of CBD products remains subject to strict regulations. While hemp-derived CBD is generally permissible, it must meet specific requirements, and shipments can be seized if they don’t comply. This adds another layer of complexity for businesses importing CBD products into the US.
Despite the federal uncertainty, state-level innovation is driving significant growth in the US CBD market. States like California, Colorado, and Oregon have established comprehensive regulatory frameworks for CBD, allowing for the sale of a wide range of products. Other states, like Texas and Florida, have more restrictive approaches.
The key differences between state laws relate to THC content limits, product testing requirements, and labeling regulations. Some states also regulate the sale of CBD in food and beverages, while others do not. This patchwork of regulations creates challenges for businesses operating in multiple states.
States with progressive CBD laws tend to have more developed markets and attract more investment. Conversely, states with restrictive laws often see slower growth and limited product availability. The ongoing debate over federal legalization is a major factor influencing the future of the US CBD market.
- Federal Prohibition: CBD remains a Schedule I controlled substance.
- 2018 Farm Bill: Legalized hemp-derived CBD with <0.3% THC.
- State-Level Innovation: California, Colorado, and Oregon have comprehensive regulations.
- Varied State Laws: Differing THC limits, testing requirements, and labeling regulations.
US CBD Regulatory Landscape
- Fully Legal - These states have comprehensive CBD regulations, generally allowing for the sale of CBD products with varying THC limits (typically below 0.3%) and robust testing requirements. Includes: California, Colorado, Oregon, Washington, Vermont, Maine, Massachusetts, Illinois, Nevada, Arizona, Connecticut, Rhode Island, New York, Maryland, Delaware, and Michigan.
- Restricted Access - These states permit CBD sales but often have specific restrictions, such as limitations on product types (e.g., only CBD oil, no edibles) or require licensing for retailers. Includes: Florida, North Carolina, Pennsylvania, Ohio, Texas, Virginia, and Minnesota.
- Limited CBD - States in this category typically allow CBD with very specific conditions, often restricting it to research purposes or requiring a doctor's recommendation. Regulations can be unclear or evolving. Includes: Georgia, South Carolina, Indiana, and Wisconsin.
- Prohibited - These states maintain outright bans on most or all CBD products, even those derived from hemp and containing less than 0.3% THC. Includes: Idaho, Kansas, and Nebraska.
- International Expansion - Germany - Germany legalized CBD products in 2022, but regulations regarding THC content and novel food status continue to evolve, impacting product availability and market access for international brands.
- International Expansion - United Kingdom - The UK's Food Standards Agency (FSA) requires CBD businesses to have novel food authorizations, creating a bottleneck for many companies and leading to market consolidation. The FSA deadline for applications was October 2023.
- International Expansion - Brazil - Brazil’s regulations around CBD are evolving. Currently, CBD products are permitted with a prescription, and there’s increasing discussion around over-the-counter availability for lower-THC products.
Asia-Pacific: Traditional Medicine and Emerging Markets
The Asia-Pacific region presents a unique opportunity for the CBD market, largely due to the long-standing acceptance of traditional medicine practices. In many countries, herbal remedies and natural health products are deeply ingrained in the culture, creating a more receptive environment for CBD.
Japan is a key market, but regulations are relatively strict. CBD products are only permitted for topical use and must be approved by the Ministry of Health, Labour and Welfare. South Korea is also a growing market, with a focus on CBD cosmetics and skincare products.
Australia has a more established regulatory framework for CBD, allowing for the sale of low-dose CBD products over the counter. However, access to higher-dose CBD products typically requires a prescription. Thailand has recently legalized CBD, opening up new opportunities for businesses.
Regulatory challenges remain a significant obstacle in many Asia-Pacific countries. Some countries have outright bans on CBD, while others have complex and unclear regulations. Navigating these regulations requires a deep understanding of local laws and customs.
CBD products that align with traditional medicine principles are particularly popular in this region. For example, CBD-infused oils and balms are often marketed as natural remedies for pain relief and inflammation. The growing middle class and increasing disposable incomes are also driving demand for CBD products.
- Traditional Medicine Acceptance: Fuels demand for natural health products.
- Japan: Strict regulations, limited to topical use.
- South Korea: Focus on CBD cosmetics and skincare.
- Australia: Low-dose CBD available over the counter.
- Thailand: Recently legalized CBD.
New Store Formats: Beyond the Traditional Retail Space
The traditional standalone CBD store is no longer the only way to buy CBD. We’re seeing a diversification of retail formats, with CBD products increasingly appearing in existing retail spaces. This is largely driven by the desire to reach a wider audience and reduce the stigma associated with CBD.
Many pharmacies and health food stores are now dedicating sections to CBD products. This provides consumers with a convenient and trusted source for CBD, and allows retailers to capitalize on the growing demand. Even some convenience stores are starting to carry CBD topicals and other products.
The growth of online CBD marketplaces and direct-to-consumer brands is another significant trend. These platforms offer a wide selection of products and often provide more competitive pricing. They also allow businesses to reach customers directly, without having to rely on traditional retail channels.
Each format has its advantages and disadvantages. Standalone stores offer a specialized shopping experience, but they can be expensive to operate. Pharmacies and health food stores benefit from existing foot traffic, but may have limited product selection. Online marketplaces offer convenience and competitive pricing, but can be challenging to navigate.
Brands are adapting to these changes by adopting a multi-channel approach, selling their products through a combination of standalone stores, retail partnerships, and online platforms. This allows them to reach a wider audience and maximize their sales potential.
Regulatory Shifts: What to Watch in 2025-2026
The next couple of years will be pivotal for the global CBD market, with several key regulatory developments on the horizon. In the United States, the push for federal legalization is gaining momentum, although the timeline remains uncertain. Any progress on this front would be a game-changer for the industry.
The EU’s review of the Novel Food regulations is also a major event to watch. We could see changes that streamline the authorization process and reduce the burden on businesses. The success of this review will largely depend on the willingness of member states to compromise and adopt a more unified approach.
In Asia-Pacific, we can expect to see further regulatory changes in countries like Thailand and Japan. Thailand’s recent legalization is likely to attract investment and spur innovation, while Japan may consider easing its restrictions on CBD use. Australia may also expand access to higher-dose CBD products.
One potential development is the increased focus on product safety and quality control. Regulators are likely to tighten standards for CBD products, requiring more rigorous testing and labeling. This is a positive development for consumers, but it could also increase compliance costs for businesses.
Businesses should be preparing for these changes by staying informed about regulatory developments, investing in quality control measures, and diversifying their product offerings. Adaptability will be key to success in this rapidly evolving market. Keeping a close watch on resources like MindCBD’s CBD directory will be invaluable.
- US Federal Legalization: Ongoing efforts, uncertain timeline.
- EU Novel Food Review: Potential for streamlined authorization.
- Asia-Pacific Regulatory Changes: Thailand and Japan to watch.
- Increased Focus on Product Safety: Tighter standards and quality control.
What do you believe is the biggest obstacle to global CBD market growth in 2026?
As the CBD industry expands into new international markets, regulatory frameworks and public understanding continue to evolve at different speeds across regions. We want to hear from our MindCBD community: which challenge do you think poses the greatest barrier to the industry reaching its full potential? Vote below and share your perspective!
MindCBD: Navigating the Expanding Market
The global CBD market is becoming increasingly complex, with new products, brands, and regulations emerging constantly. Navigating this landscape can be challenging for both consumers and businesses. That's where MindCBD comes in.
MindCBD is a comprehensive directory of global CBD stores, providing a valuable resource for anyone looking to buy or sell CBD products. Our CBD store locator allows you to find nearby stores, while our detailed brand profiles offer insights into product quality and reputation.
We also offer in-depth product reviews, helping consumers make informed purchasing decisions. Our team of experts carefully evaluates CBD products based on factors like potency, purity, and third-party testing. We aim to provide unbiased and reliable information.
For businesses, MindCBD offers a platform to showcase their products and reach a wider audience. We provide marketing and advertising opportunities to help businesses grow their brand awareness and drive sales. It’s a way to connect with a targeted audience actively seeking CBD solutions.
Ultimately, MindCBD is committed to providing a trusted resource for the CBD community. We believe that transparency and education are essential for fostering a sustainable and responsible CBD market.




No comments yet. Be the first to share your thoughts!